

Paraformaldehyde, also known as PFA, is the linear polymer of formaldehyde, typically appearing as a white solid powder or granules. It does not have a fixed melting point but depolymerizes upon heating within a certain temperature range, releasing gaseous formaldehyde. Compared to formaldehyde aqueous solution (formalin), Paraformaldehyde offers significant advantages such as high purity, extremely low water content, good stability, and ease of transportation and storage. It serves as an important and convenient “solid formaldehyde” source.It is also a chemical intermediate in modern chemical production.Paraformaldehyde is mainly used in biological research as a fixative.It preserves tissue and cell structures for microscopy. Industrially, it serves as a crucial raw material for manufacturing synthetic resins and plastics. Additionally, it functions as a disinfectant and fumigant,
| CAS: | 30525-89-4 | UN: | 2213 |
| Other Names: | Polyoxymethylene, Formaldehyde polymer, Formagene | Appearance: | It appears as a white amorphous powder, odorless or slightly with a pungent odor of formaldehyde. |
| Density: | 1.46 g/cm³ | Melting point: | 120 °C |
| Boiling point: | °C | Flash point: | 71°C |
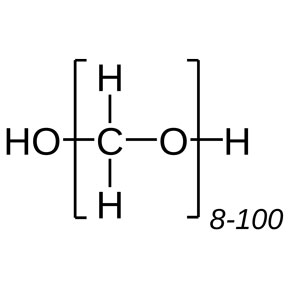
| Molecular Formula: | OH(CH2O)nH | ||












